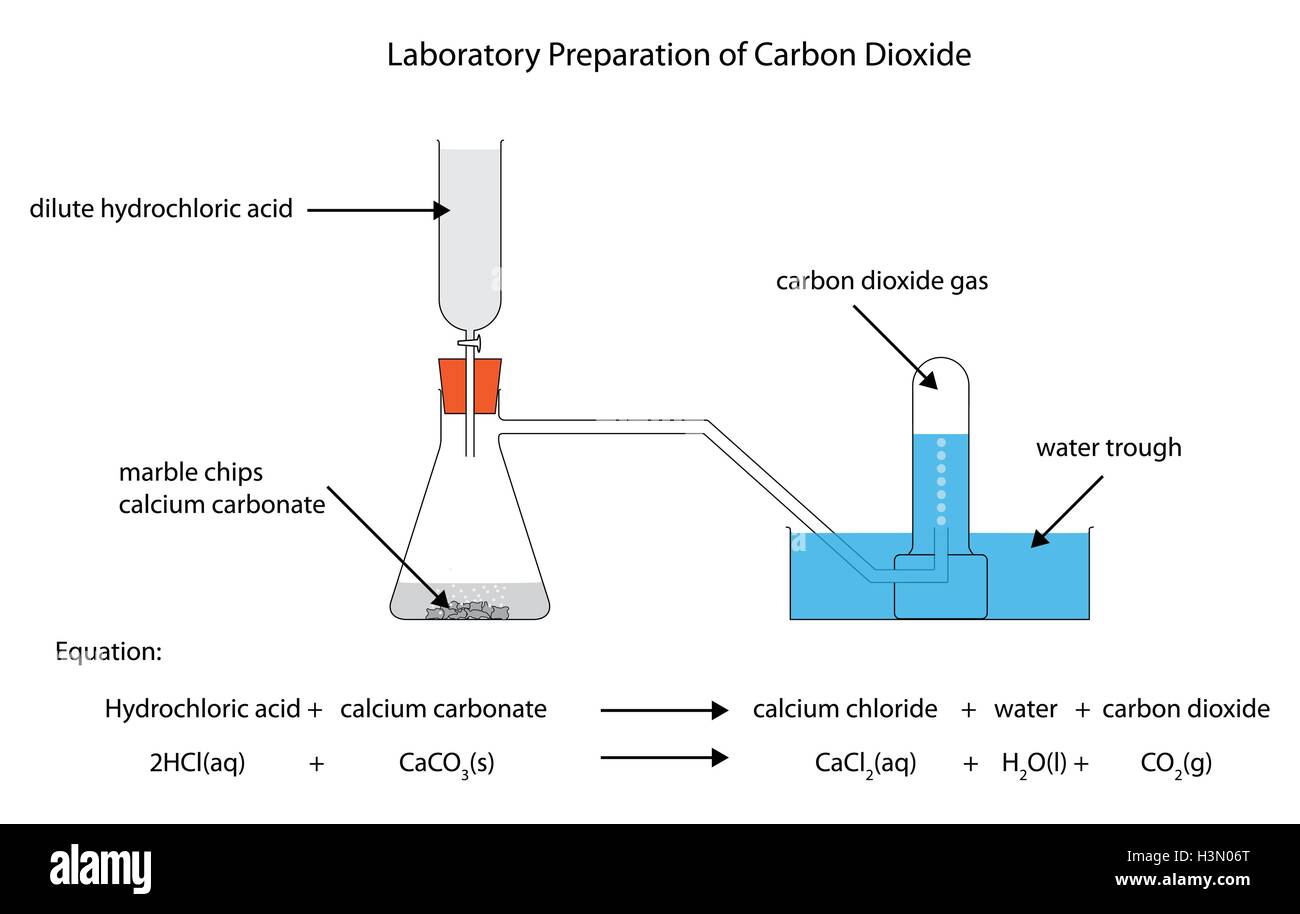
Diagram of the laboratory preparation of carbon dioxide from hydrochloric acid and calcium carbonate marble chips Stock Vector Image & Art - Alamy

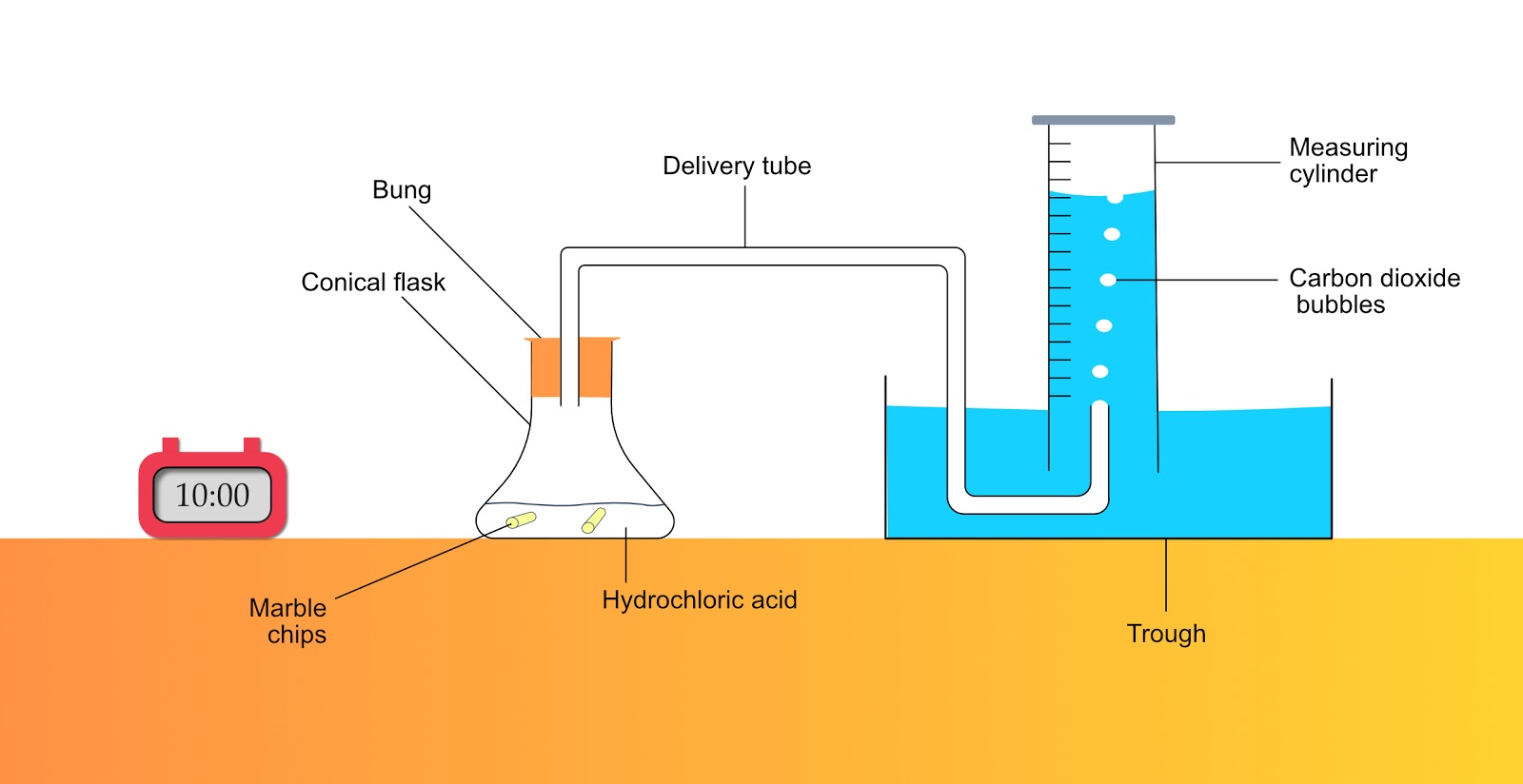
Powder marble reacts with hydrochloric acid using the apparatus shown.The gas syringe fills in 36 seconds.The experiment is repeated using marble chips in place of powdered marble.How long it take to fill

a student was investigating the reaction between marble chips and dilute hydrochloric acid - Brainly.com

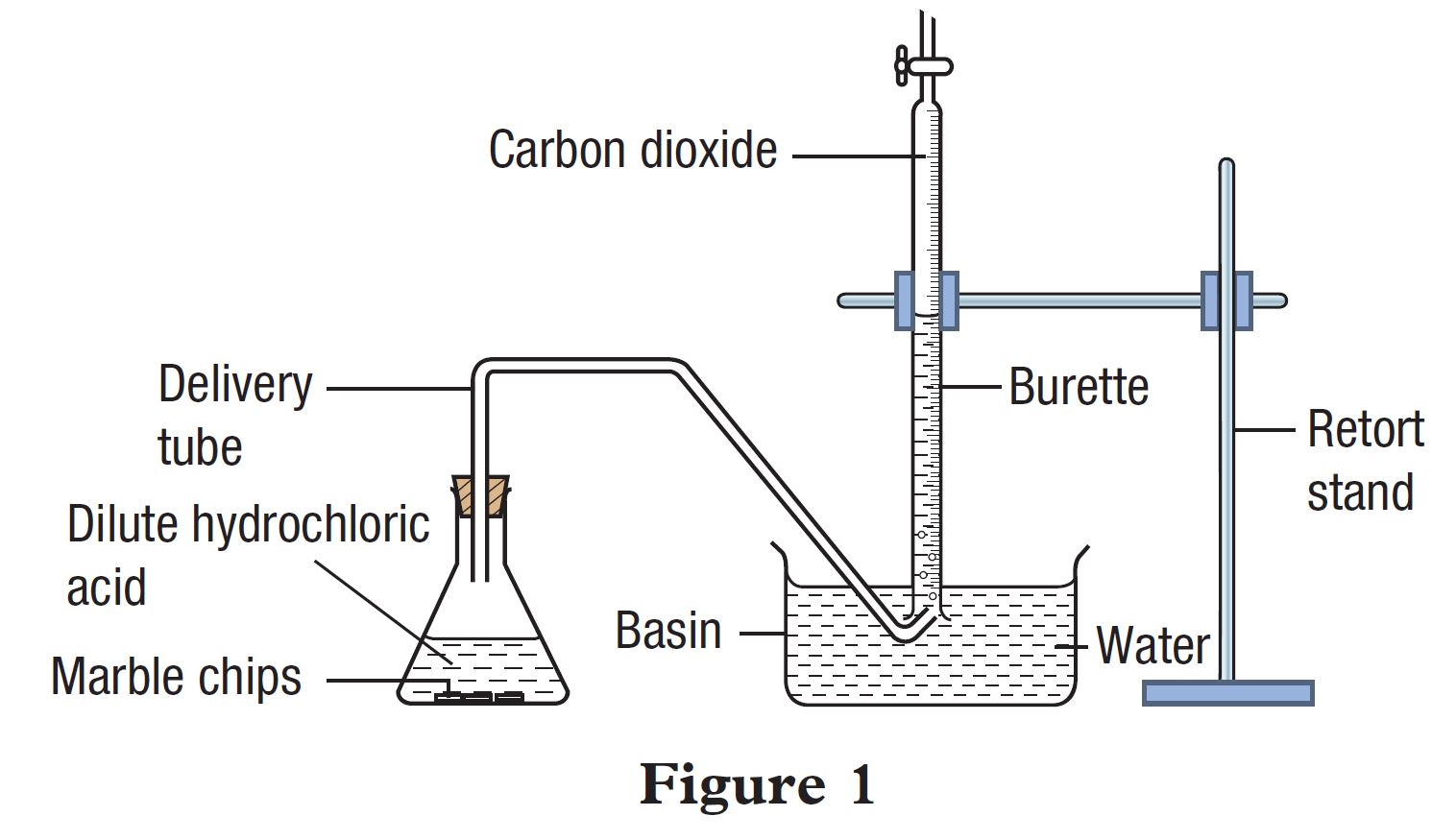
investigate the effect of changing the surface area of marble chips and of changing the concentration of hydrochloric acid on the rate of reaction between marble chips and dilute hydrochloric acid Diagram
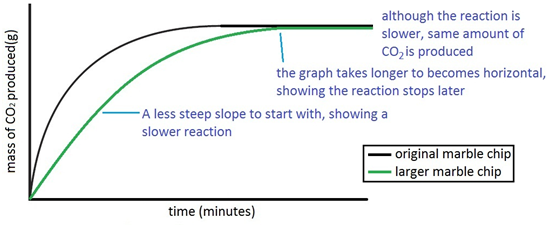
3:15 practical: investigate the effect of changing the surface area of marble chips and of changing the concentration of hydrochloric acid on the rate of reaction between marble chips and dilute hydrochloric

3:15 practical: investigate the effect of changing the surface area of marble chips and of changing the concentration of hydrochloric acid on the rate of reaction between marble chips and dilute hydrochloric
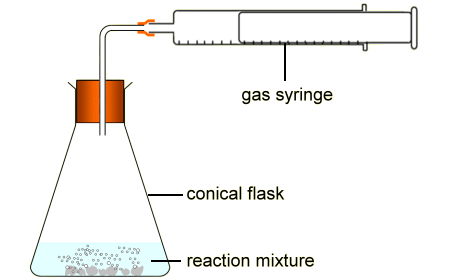
Chemistry Rate of Reaction Coursework for Calcium Carbonate and Hydrochloric Acid - GCSE Science - Marked by Teachers.com

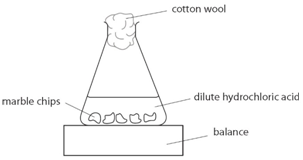
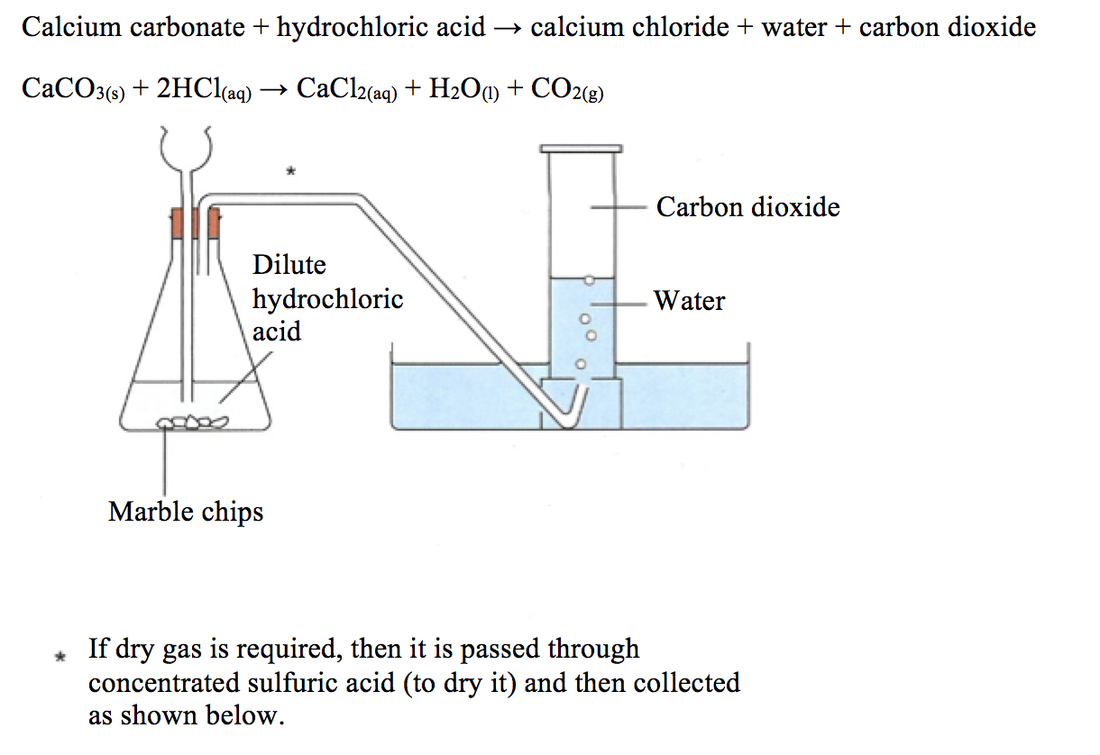
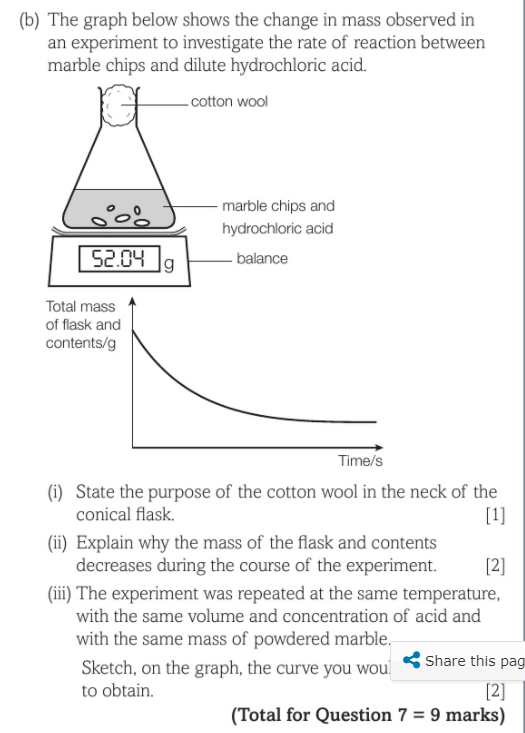
Describe an experiment to study the speed of reaction between calcium carbonate and dilute hydrochloric acid, by measuring the loss in mass of reaction system over time. - Study notes, tips, worksheets,