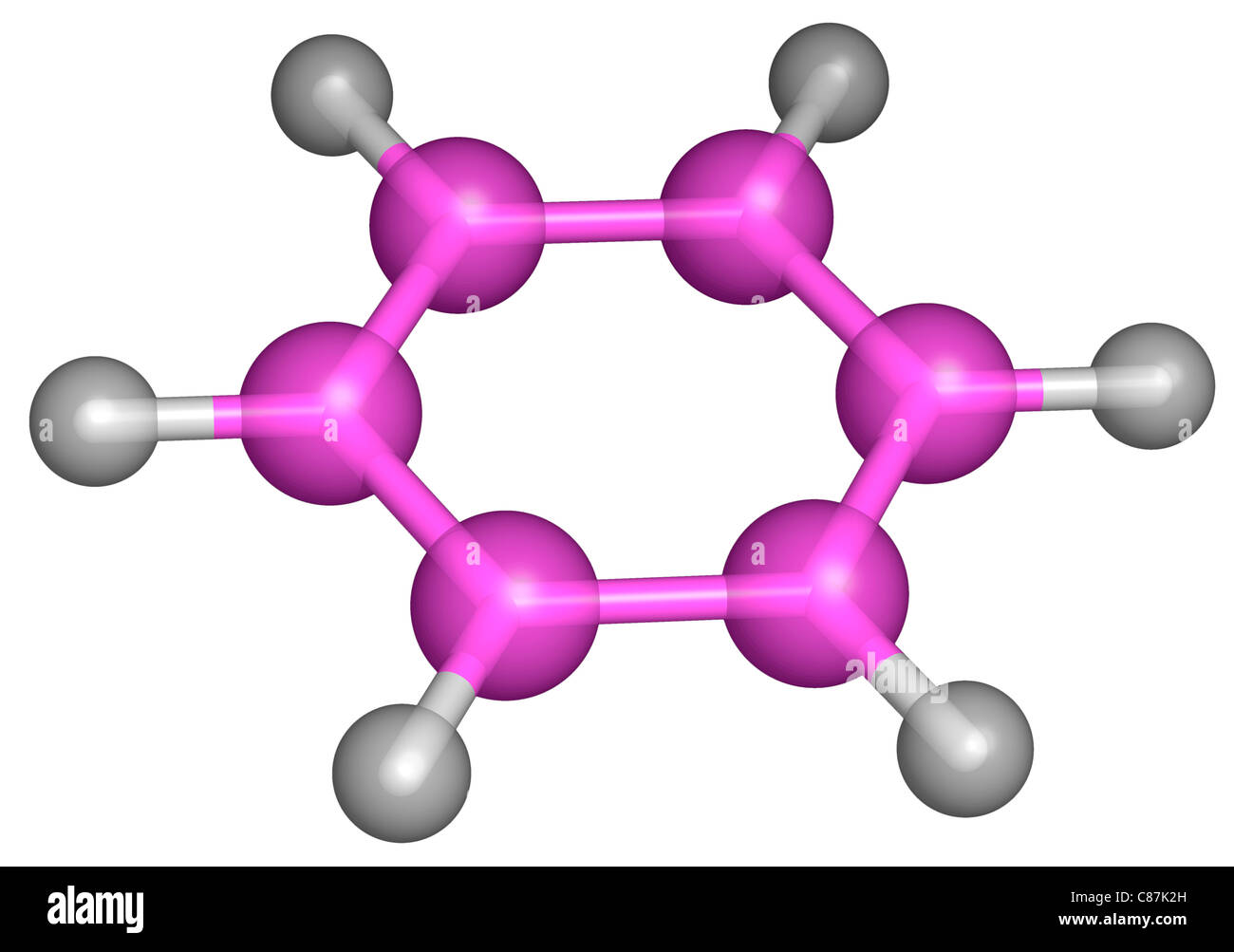
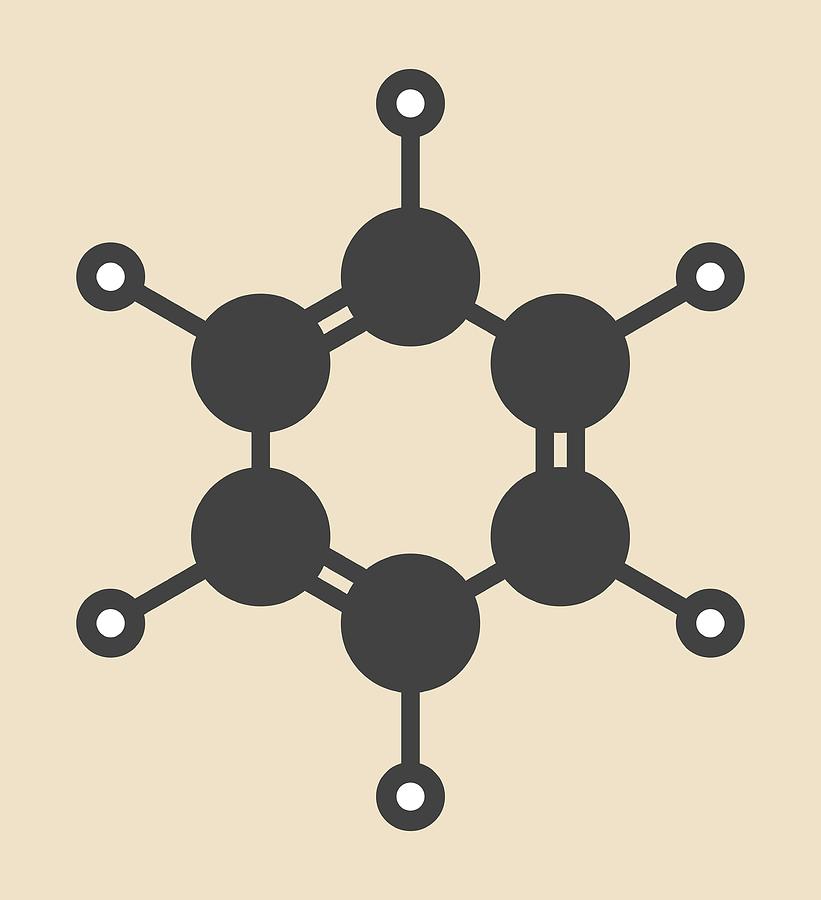
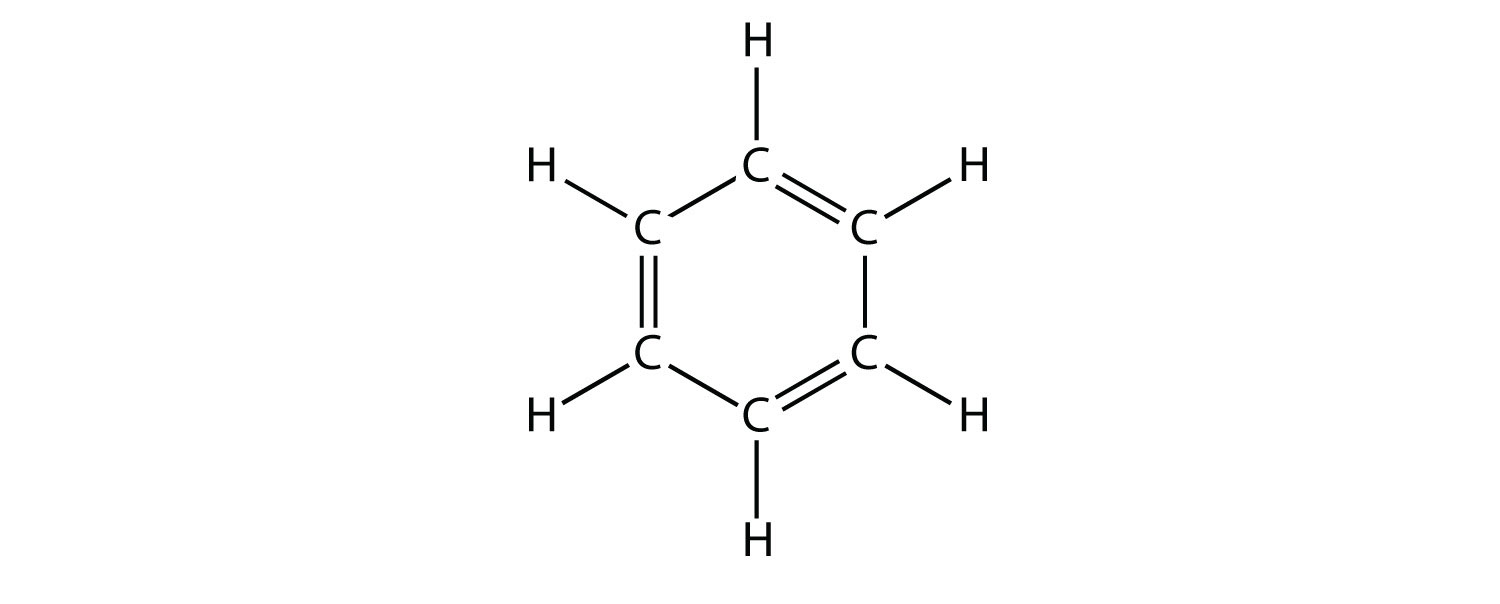
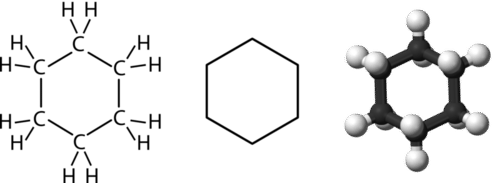
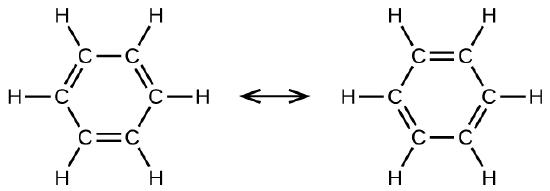
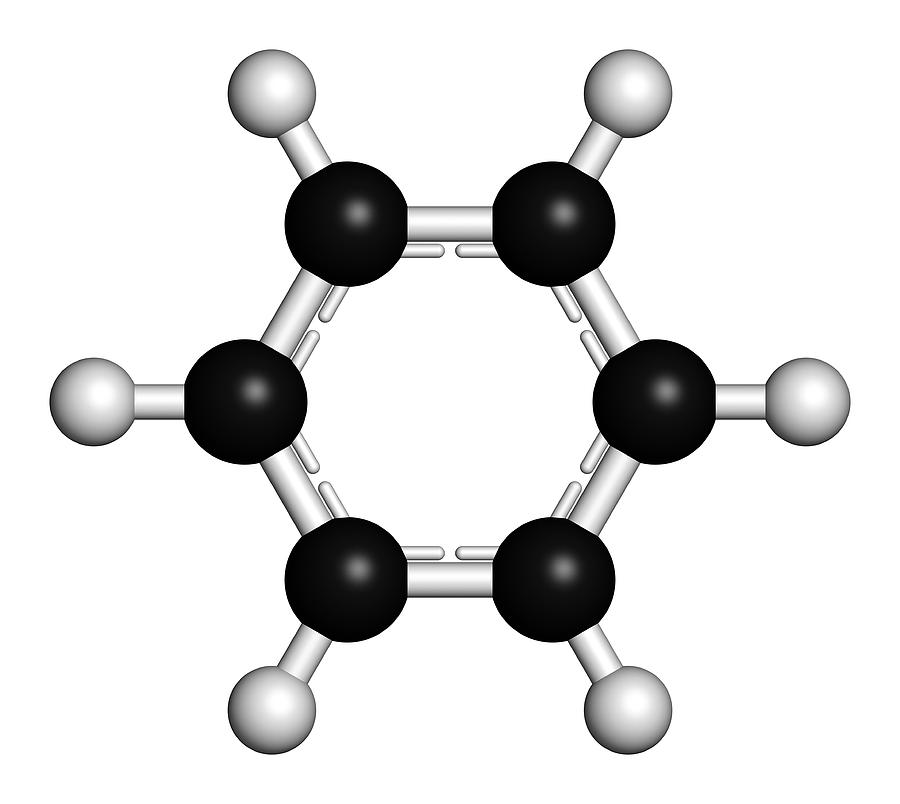
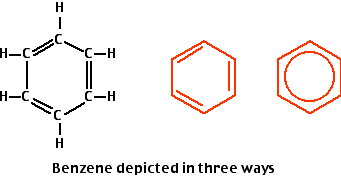
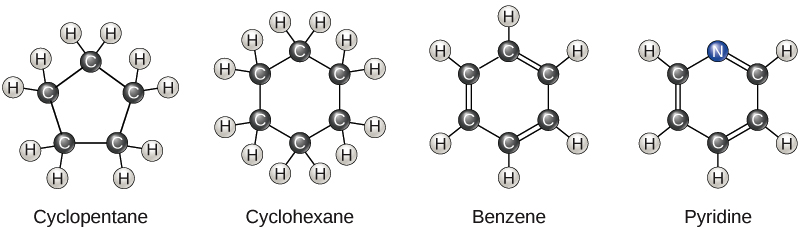
What are aromatic hydrocarbons? Give a detailed description of the bonding in benzene. The pi electrons in benzene are delocalized, whereas the pi electrons in simple alkenes and alkynes are localized.

Molecule Chemistry Aromatic hydrocarbon Benzene, aromatic ring, biology, chemistry, acid png | PNGWing
![Corannulene is a polycyclic aromatic hydrocarbon with chemical formula C20H10.[1] The molecule consists of a cyclopentane ring fused with 5 benzene ri Stock Photo - Alamy Corannulene is a polycyclic aromatic hydrocarbon with chemical formula C20H10.[1] The molecule consists of a cyclopentane ring fused with 5 benzene ri Stock Photo - Alamy](https://c8.alamy.com/comp/2BJHN82/corannulene-is-a-polycyclic-aromatic-hydrocarbon-with-chemical-formula-c20h10-1-the-molecule-consists-of-a-cyclopentane-ring-fused-with-5-benzene-ri-2BJHN82.jpg)
Corannulene is a polycyclic aromatic hydrocarbon with chemical formula C20H10.[1] The molecule consists of a cyclopentane ring fused with 5 benzene ri Stock Photo - Alamy

A hydrocarbon consists of a benzene ring and two methyl groups. The methyl groups are located at opposite carbons and the molecule is perfectly symmetrical. Which option correctly describes the numbers of