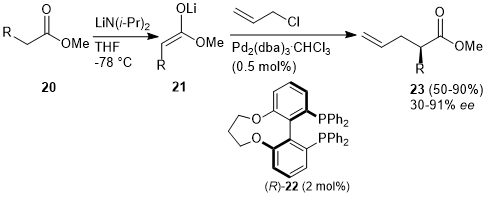
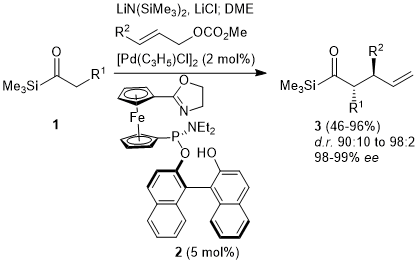
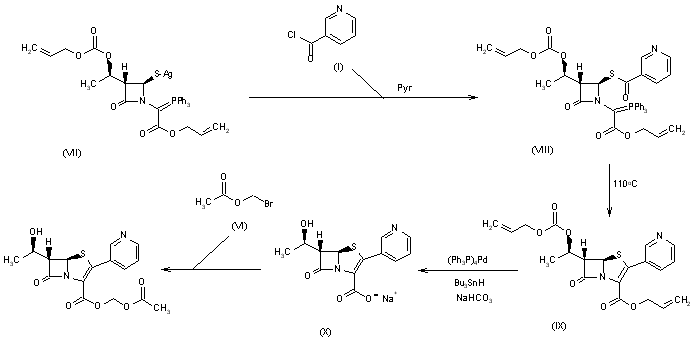
Cobalt-Catalyzed Deprotection of Allyl Carboxylic Esters Induced by Hydrogen Atom Transfer | Organic Letters

Mechanism of allyl deprotection through catalytic palladium π-allyl... | Download Scientific Diagram
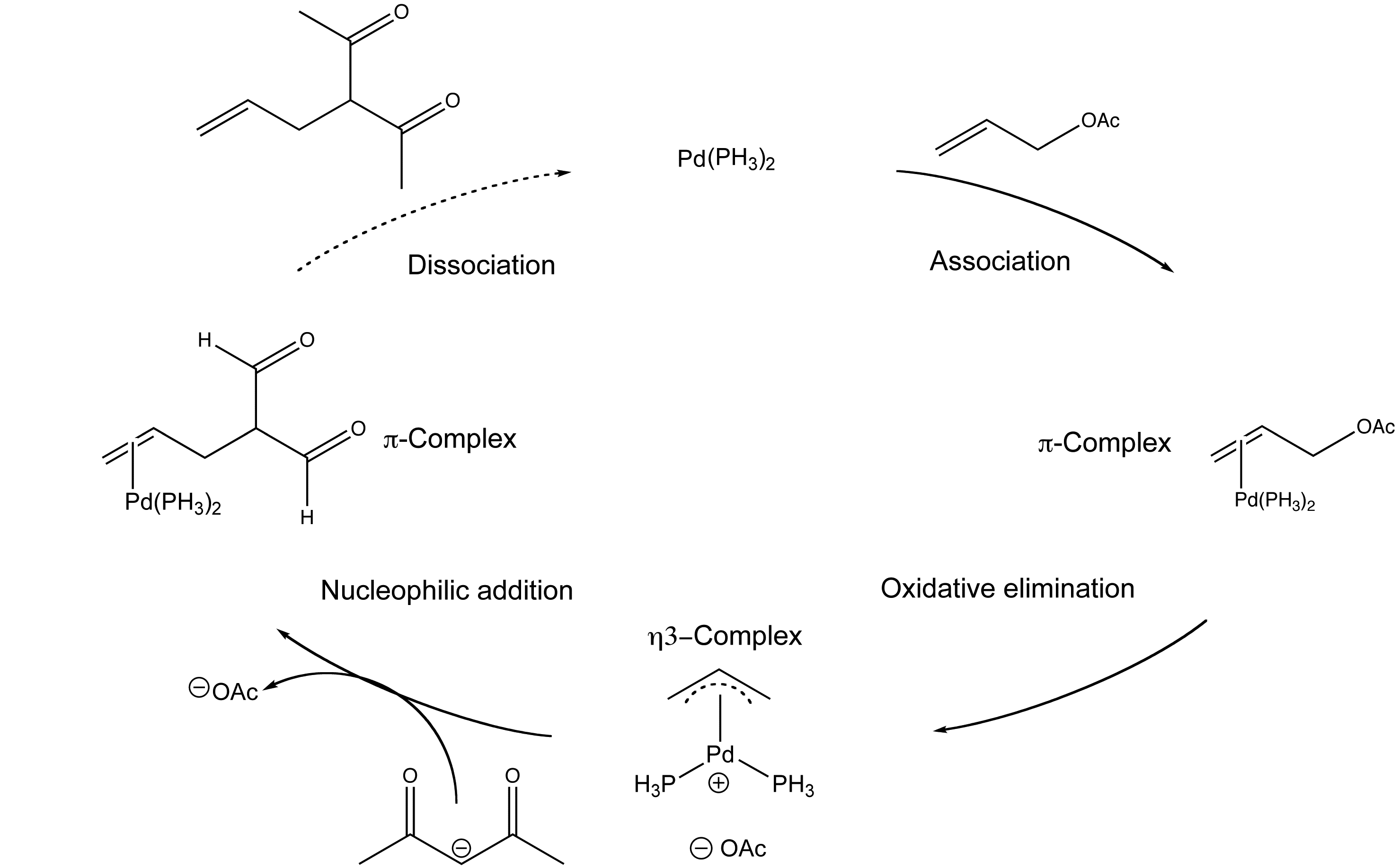
Organopalladium Chemistry - Palladium-catalysed nucleophilic allylic substitution of functionalised compounds

Facile and selective cleavage of allyl ethers, amines and esters using polymethylhydrosiloxane–ZnCl2/Pd(PPh3)4 - ScienceDirect

Irreversible Catalytic Ester Hydrolysis of Allyl Esters to Give Acids and Aldehydes by Homogeneous Ruthenium and Ruthenium/Palladium Dual Catalyst Systems - Nakamura - 2011 - Advanced Synthesis & Catalysis - Wiley Online Library

Palladium(0)‐Catalyzed Rearrangement of Allylic Esters - Jessen - 2020 - ChemistrySelect - Wiley Online Library
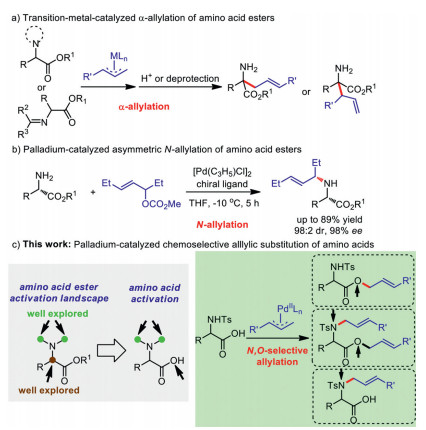
Palladium-catalyzed base- and solvent-controlled chemoselective allylation of amino acids with allylic carbonates

Amide α,β-Dehydrogenation Using Allyl-Palladium Catalysis and a Hindered Monodentate Anilide. | Semantic Scholar
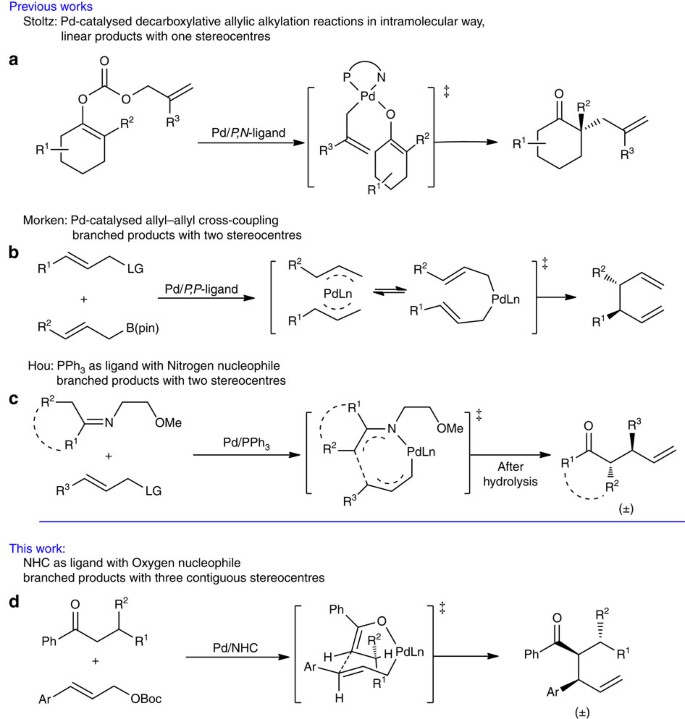
Palladium/N-heterocyclic carbene catalysed regio and diastereoselective reaction of ketones with allyl reagents via inner-sphere mechanism | Nature Communications
![PDF] Palladium-catalyzed enantioselective decarboxylative allylic alkylation of fully substituted N-acyl indole-derived enol carbonates† †Electronic supplementary information (ESI) available: Experimental procedures, NMR and IR spectra, SFC traces, X ... PDF] Palladium-catalyzed enantioselective decarboxylative allylic alkylation of fully substituted N-acyl indole-derived enol carbonates† †Electronic supplementary information (ESI) available: Experimental procedures, NMR and IR spectra, SFC traces, X ...](https://d3i71xaburhd42.cloudfront.net/79f141ae780dcc87caaf3d4902b4d8e8c807f10c/2-Figure1-1.png)
PDF] Palladium-catalyzed enantioselective decarboxylative allylic alkylation of fully substituted N-acyl indole-derived enol carbonates† †Electronic supplementary information (ESI) available: Experimental procedures, NMR and IR spectra, SFC traces, X ...
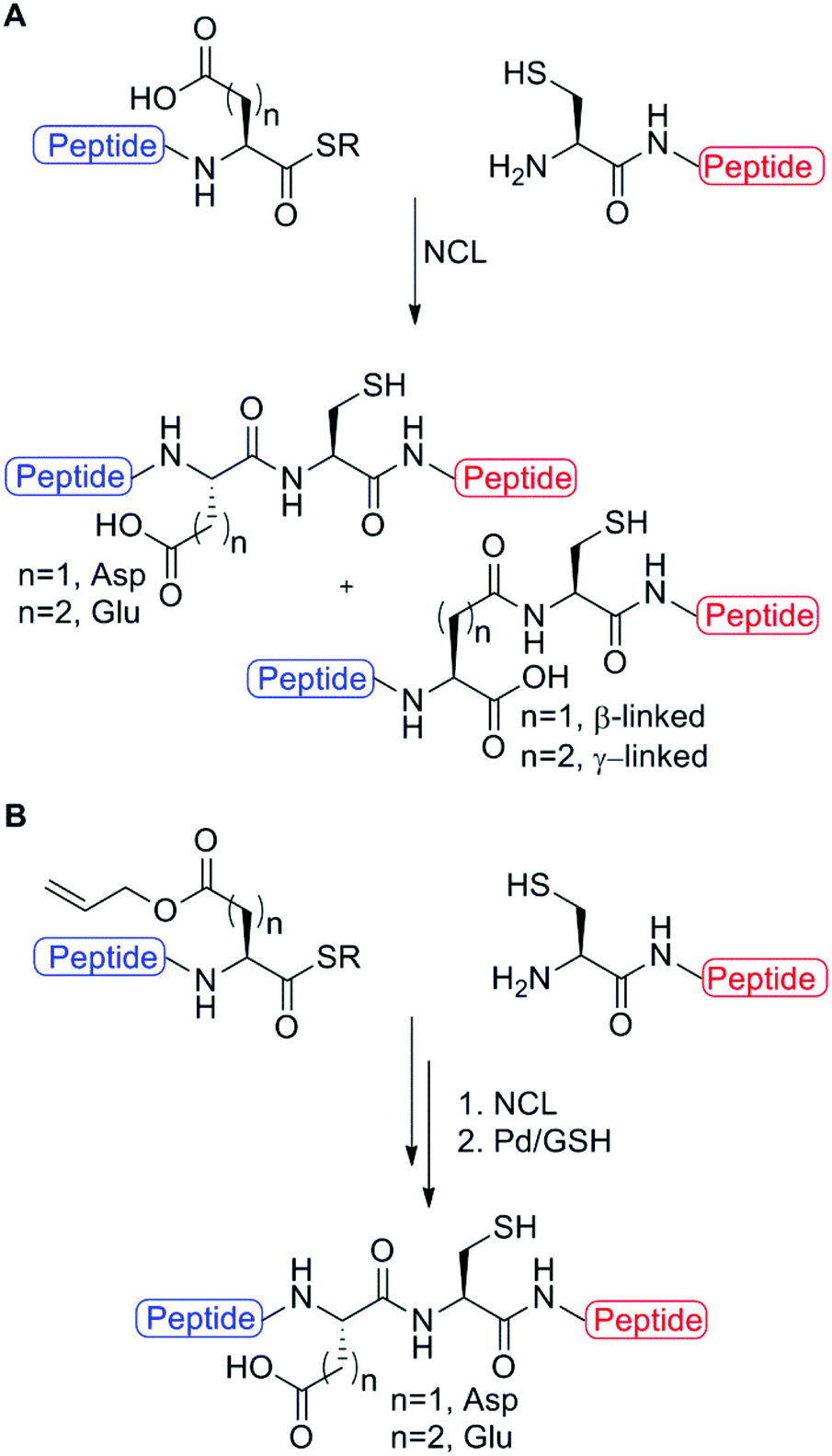
Palladium mediated deallylation in fully aqueous conditions for native chemical ligation at aspartic and glutamic acid sites - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB00890F